WSF OpenID Connect Login
KYSS – The Kyleena Satisfaction Study - provides real-world evidence on Kyleena® satisfaction.
The study confirms high satisfaction rates of women using Kyleena®, as well as a favorable placement experience by the HCP. You can find more detailed information on KYSS below. Use the tabs to scroll through insights on the relevance of Real World Evidence, the study, the HCP and patient perspective as well as safety information.
Title
KYSS: providing valuable insights into Kyleena® satisfaction
Women’s satisfaction is an important aspect in ensuring contraceptive continuation.1 Although widely used, methods such as COCs or condoms are associated with poor compliance and continuation, which can put women at risk of unintended pregnancies.2,3
Recent years have seen IUS increase in popularity among both HCPs and women,1,4,5 giving added importance to real-world studies into IUS satisfaction.
The Kyleena® Satisfaction Study (KYSS) is the first study to assess women’s satisfaction with Kyleena® in a real-world setting.6 Its insights into Kyleena® satisfaction can help to address the misperceptions around IUS use, particularly in nulliparous women, and help shape your conversations with patients who would benefit from IUS contraception.
Highlight the high satisfaction rates from KYSS to your patients during contraceptive counselling
COC – combined oral contraceptive; HCP – healthcare professional; IUS – intrauterine system; KYSS – Kyleena® Satisfaction Study
- Merki-Feld GS et al. Eur J Contracept Reprod Health Care 2018;23:183–193. Return to content
- Brynhildsen J. Ther Adv Drug Saf 2014;5(5):201–213. Return to content
- Pinter B. Eur J Contracept Reprod Health Care 2002;7(3):178–183. Return to content
- Gemzell-Danielsson K et al. Contraception 2012;86(6):631–638. Return to content
- Nelson AL and Massoudi N. Open Access J Contracept 2016;7:127–141. Return to content
- Beckert V et al. Eur J Contracept Reprod Health Care 2020;25:182–189. Return to content
Title
A real-world perspective on Kyleena®
Real-world studies are able to cover the diversity of patients and clinical scenarios seen in routine clinical practice, providing insights that are impossible to obtain during clinical trials.1,2
KYSS was designed to assess real-world evidence on Kyleena® satisfaction.3
KYSS – Kyleena® Satisfaction Study
- Berger ML et al. Value in Health 2017;20(8):1003–1008. Return to content
- Heikinheimo O et al. Eur J Contracept Reprod Health Care 2017;22:250–259. Return to content
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
- Gemzell-Danielsson K et al. Contraception 2012;86(6):631–638. Return to content
- Nelson AL and Massoudi N. Open Access J Contracept 2016;7:127–141. Return to content
- Merki-Feld GS et al. Eur J Contracept Reprod Health Care 2018;23:183–193. Return to content
Title
KYSS: one of the most recent real-world studies in the field of contraception
Title
Key study details:1
- KYSS is a prospective, multinational, single-arm, observational study
- Kyleena® placement was attempted in 1,129 women and successful in 1,126 women
- 8 countries were enrolled
- 51.9% (n=584) of women were nulliparous
- 67.1% (n=756) were 18–35 years of age
- 32.9% (n=371) of women used an oral HC as their previous method*
KYSS: designed to reflect the patients you are likely to see in your daily practice1
- Either a COC or a POP.1 Return to content
COC – combined oral contraceptive; HC – hormonal contraception; KYSS – Kyleena® Satisfaction Study;
POP – progestogen-only pill
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
Want to explore KYSS data in more detail? The final study results are available for download
A recent real-world study in the field of contraception, KYSS, has provided valuable insight into women’s and HCPs’ experience with Kyleena®.1 You can download the final study results and explore the data in detail.
HCP – healthcare professional; KYSS – Kyleena® Satisfaction Study
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
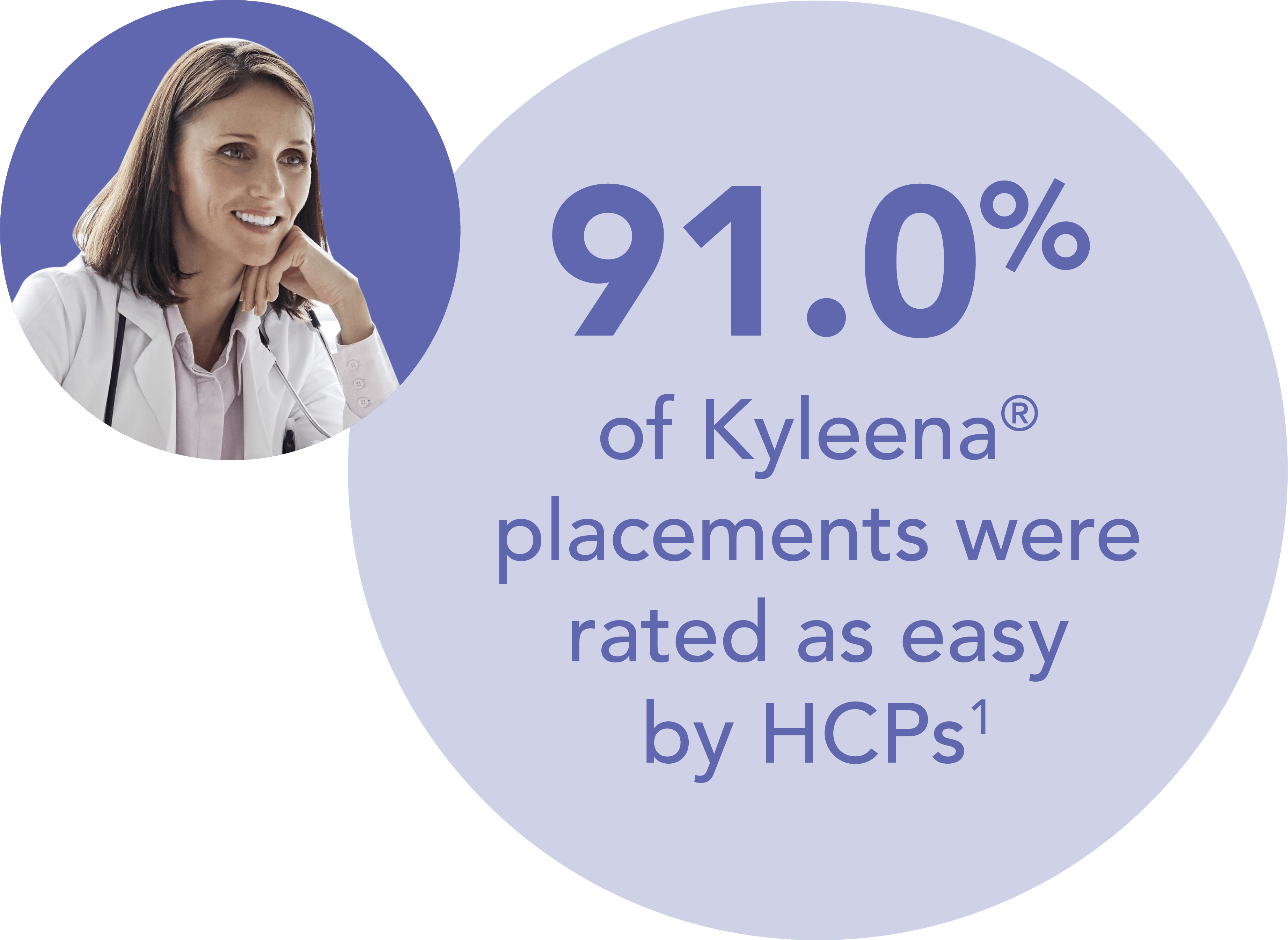
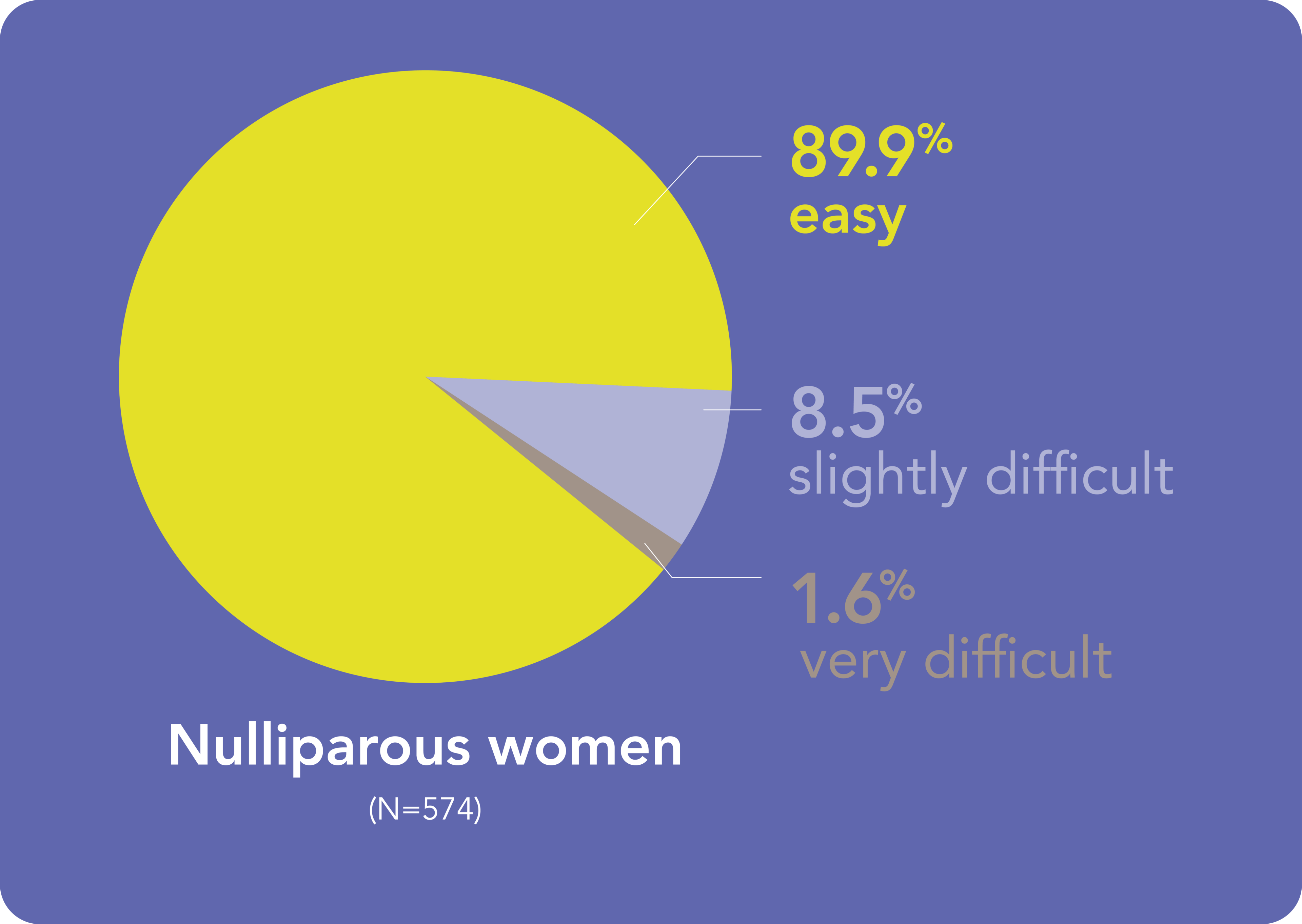
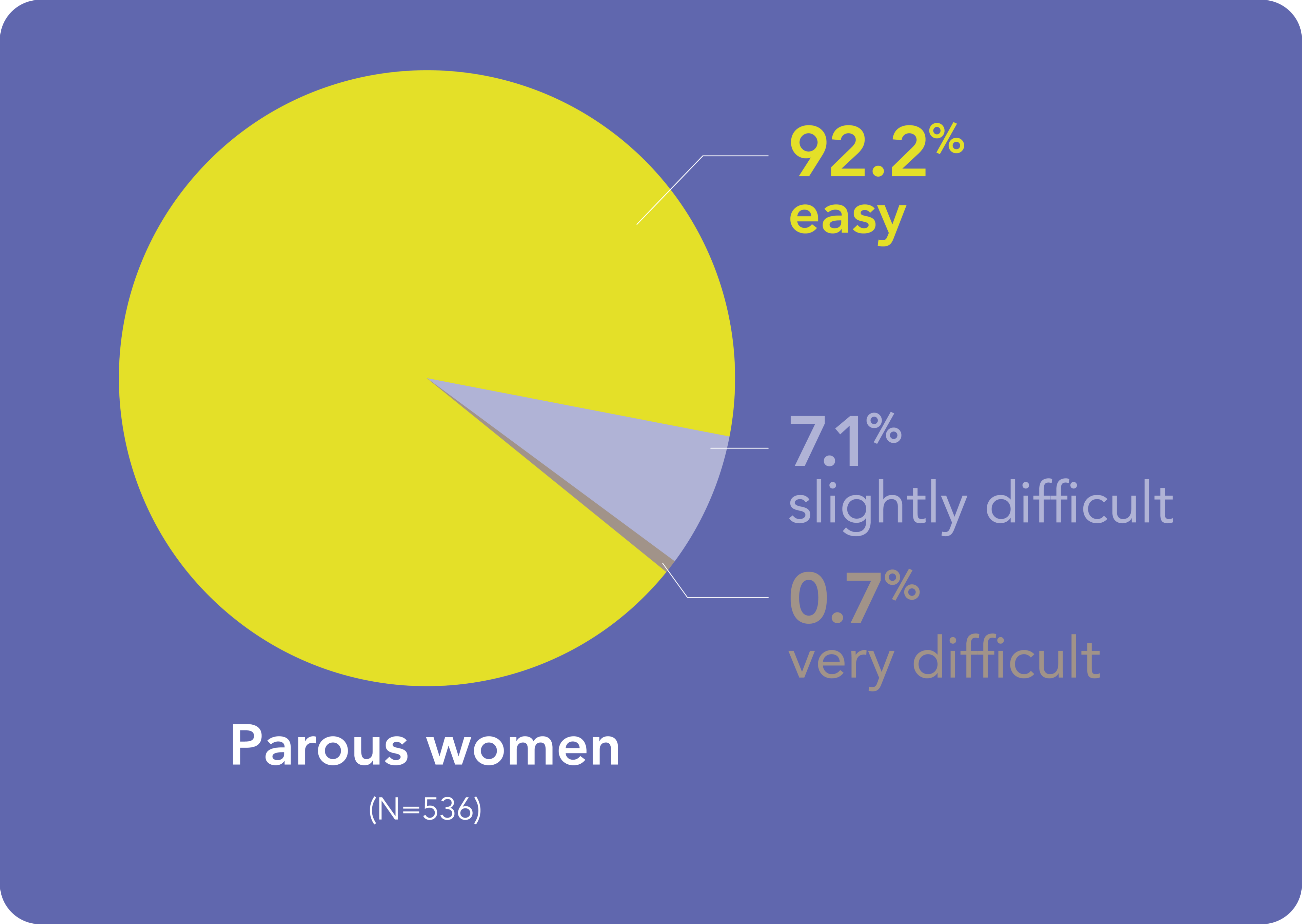
You can expect most Kyleena® placements to be straightforward, irrespective women’s parity
- Results from a baseline analysis of the KYSS data, conducted to evaluate demographics, ease of placement assessed by investigators, pain at placement rated by women, additional interventions for placement, and adverse events. A total of 1,110 women (574 nulliparous, 536 parous) from 7 countries were included in this analysis.1 Return to content
HCP – healthcare professional; KYSS – Kyleena® Satisfaction Study
- Beckert V et al. Eur J Contracept Reprod Health Care 2020;25:182–189. Return to content
Title
You can counsel your patients interested in Kyleena® with real-world satisfaction data from KYSS
KYSS provided real-world insight into women’s Kyleena® satisfaction, based on their parity, age, previous contraception, motivation for choosing Kyleena®, and country of residence.1
KYSS – Kyleena® Satisfaction Study
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
KYSS: women’s motivations for choosing Kyleena® go beyond reliable efficacy1
While high contraceptive reliability is important, women have other reasons for choosing Kyleena®.
Title
Women’s top four reasons for choosing Kyleena®:1
- No daily, weekly, or monthly contraceptive routine: 34.3% (n=386)
- High contraceptive reliability: 26.8% (n=302)
- Low-hormone dose: 25.9% (n=292)
- Expectation of shorter, lighter, and less frequent bleeding episodes: 18.6% (n=209)
Women select Kyleena® for a number of reasons. Consider counselling them on how Kyleena® can meet their individual contraceptive needs
KYSS – Kyleena® Satisfaction Study
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
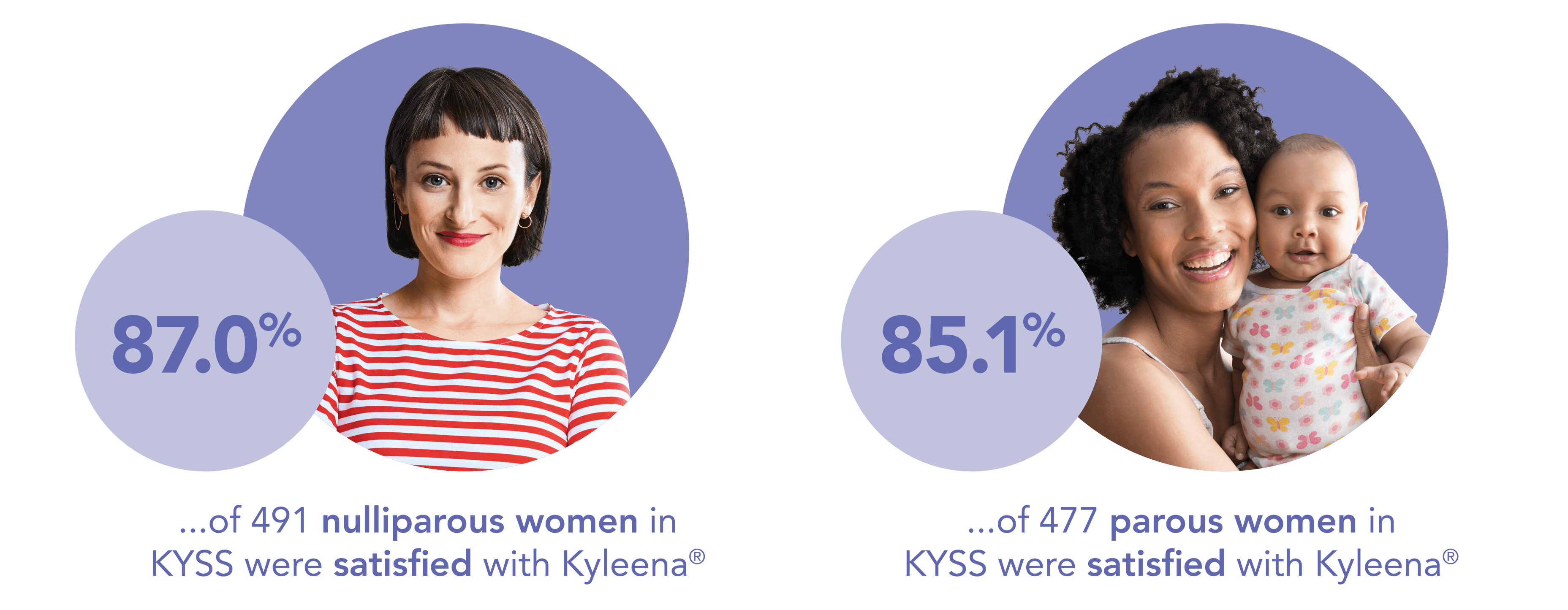
Kyleena®: high satisfaction rates, regardless of parity and age1
Satisfaction was similarly high for both parous and nulliparous women*1
Title

Reassure your patients that parity and age should not impact Kyleena® satisfaction
- Results shown represent women who reported being “very satisfied” or “somewhat satisfied” with Kyleena® at the end of the final visit.1 Return to content
KYSS – Kyleena® Satisfaction Study
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
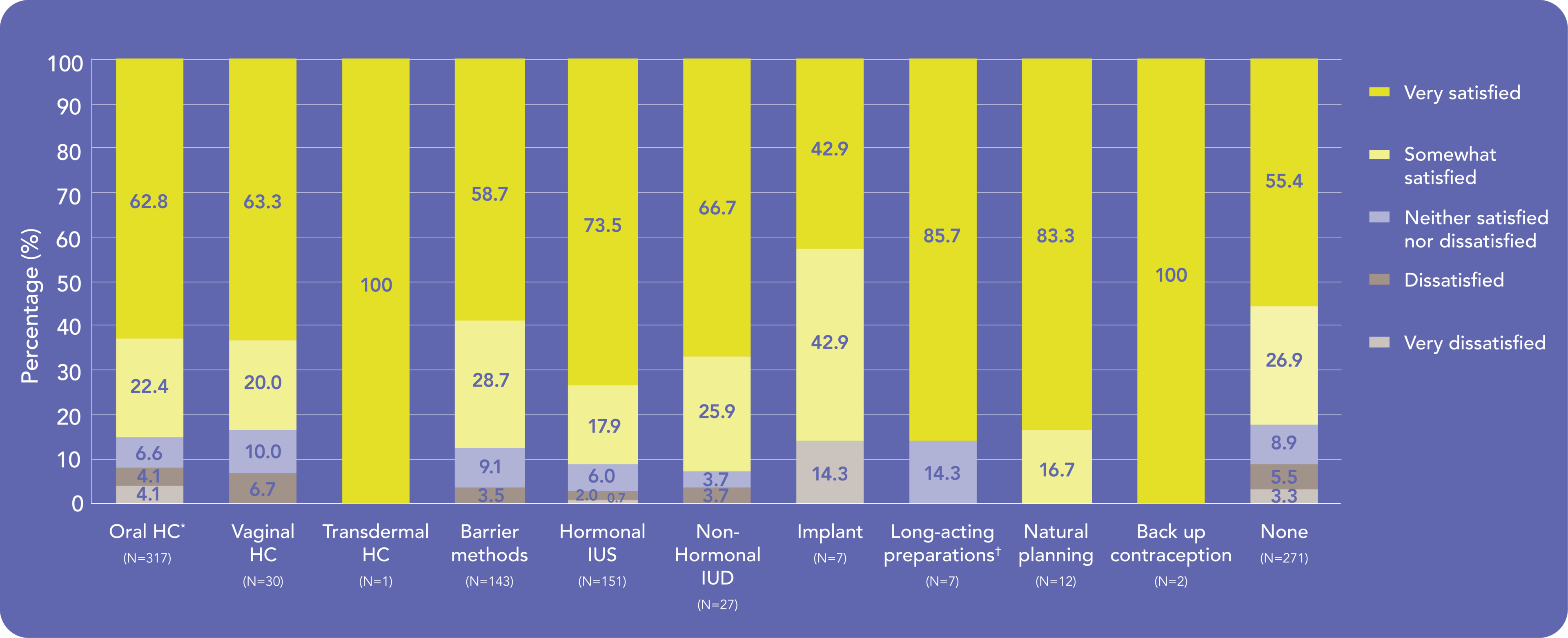
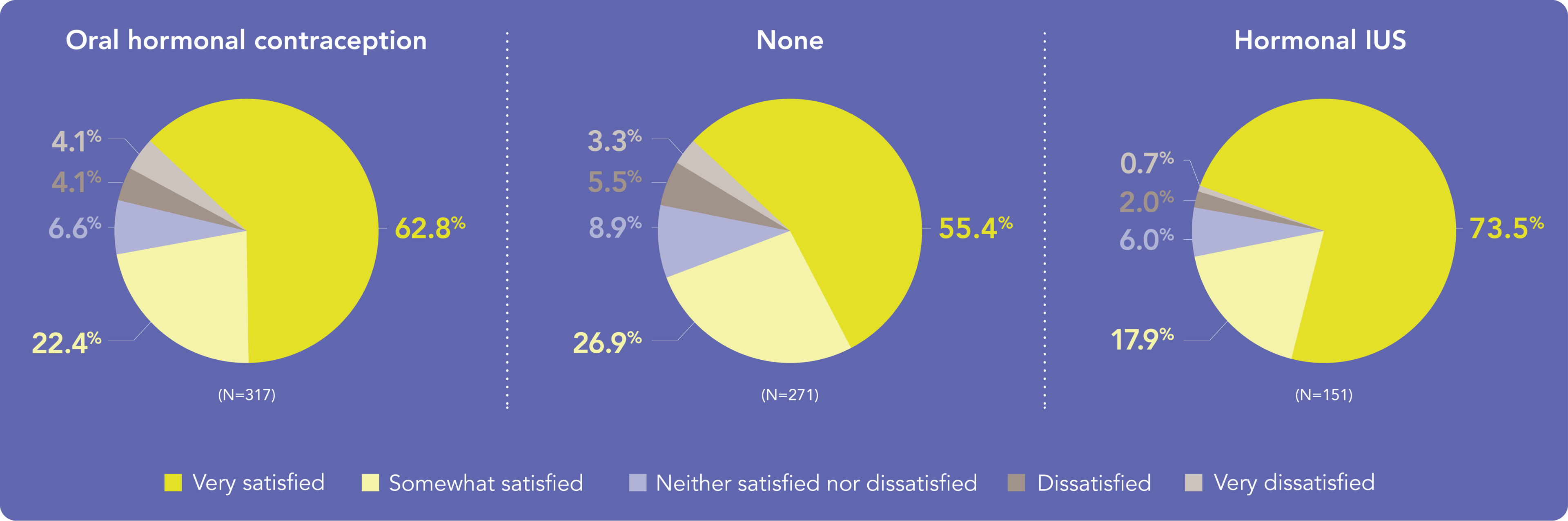
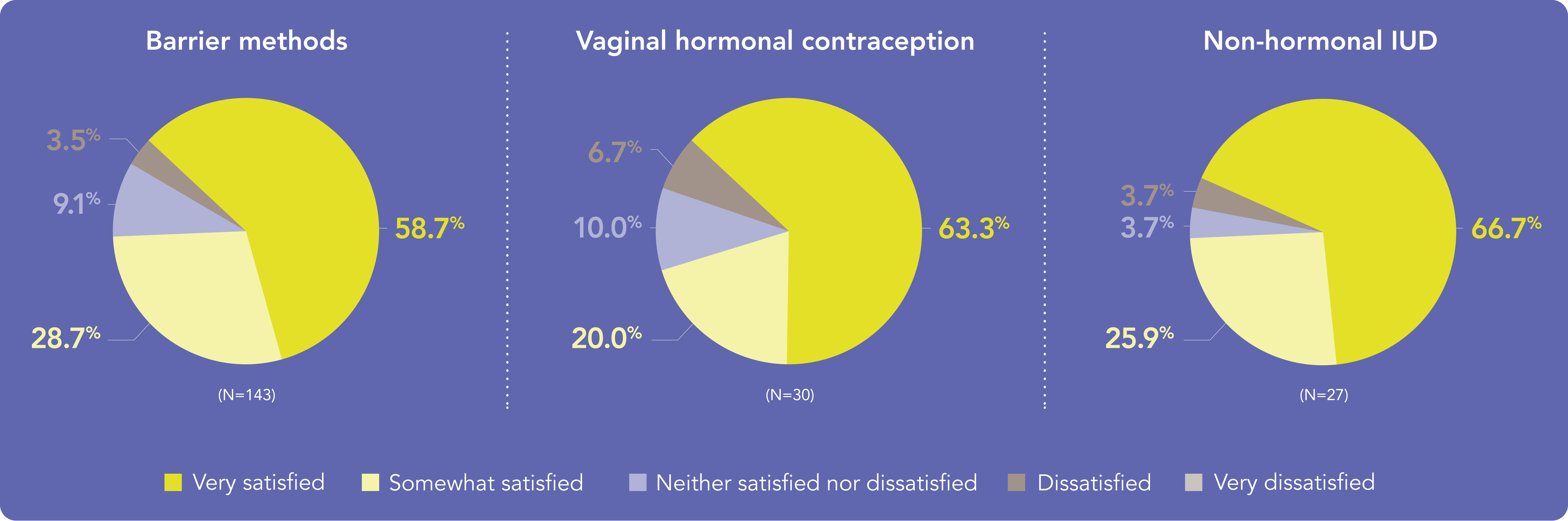
Most women were satisfied with Kyleena®, no matter their…1
…Previous contraception:
Title
…Motivation for choosing Kyleena®:
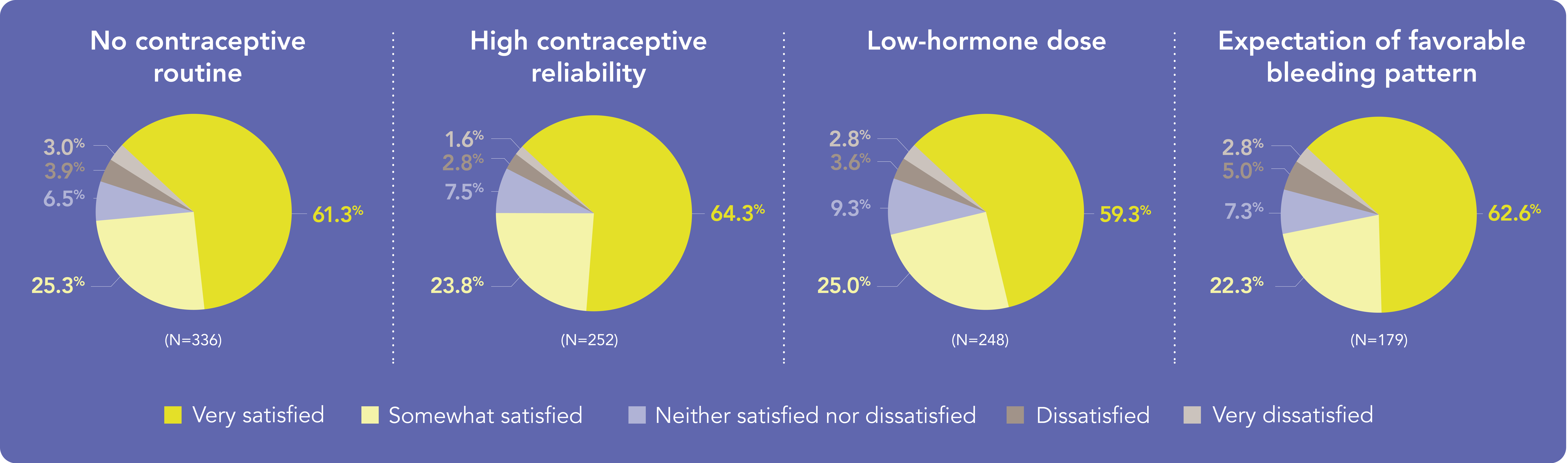
Discuss how Kyleena® meets your patients’ needs no matter their previous contraception and motivation for choosing Kyleena®
IUD – intrauterine device; IUS – intrauterine system
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
Kyleena® demonstrated a high level of satisfaction with the bleeding profile in KYSS1
Bleeding profile satisfaction is important, as it can significantly impact women’s quality of life.2-4

Discuss the high level of bleeding profile satisfaction seen in KYSS with your patients
KYSS – Kyleena® Satisfaction Study
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
- Schoep ME et al. BMJ Open 2019;9(6):026186. Return to content
- Schoep ME et al. AM J Obstet Gynecol 2019;220(6):569. Return to content
- Sveinsdóttir H. J Clin Nurs 2018;27(3–4):e503–e513. Return to content
Title
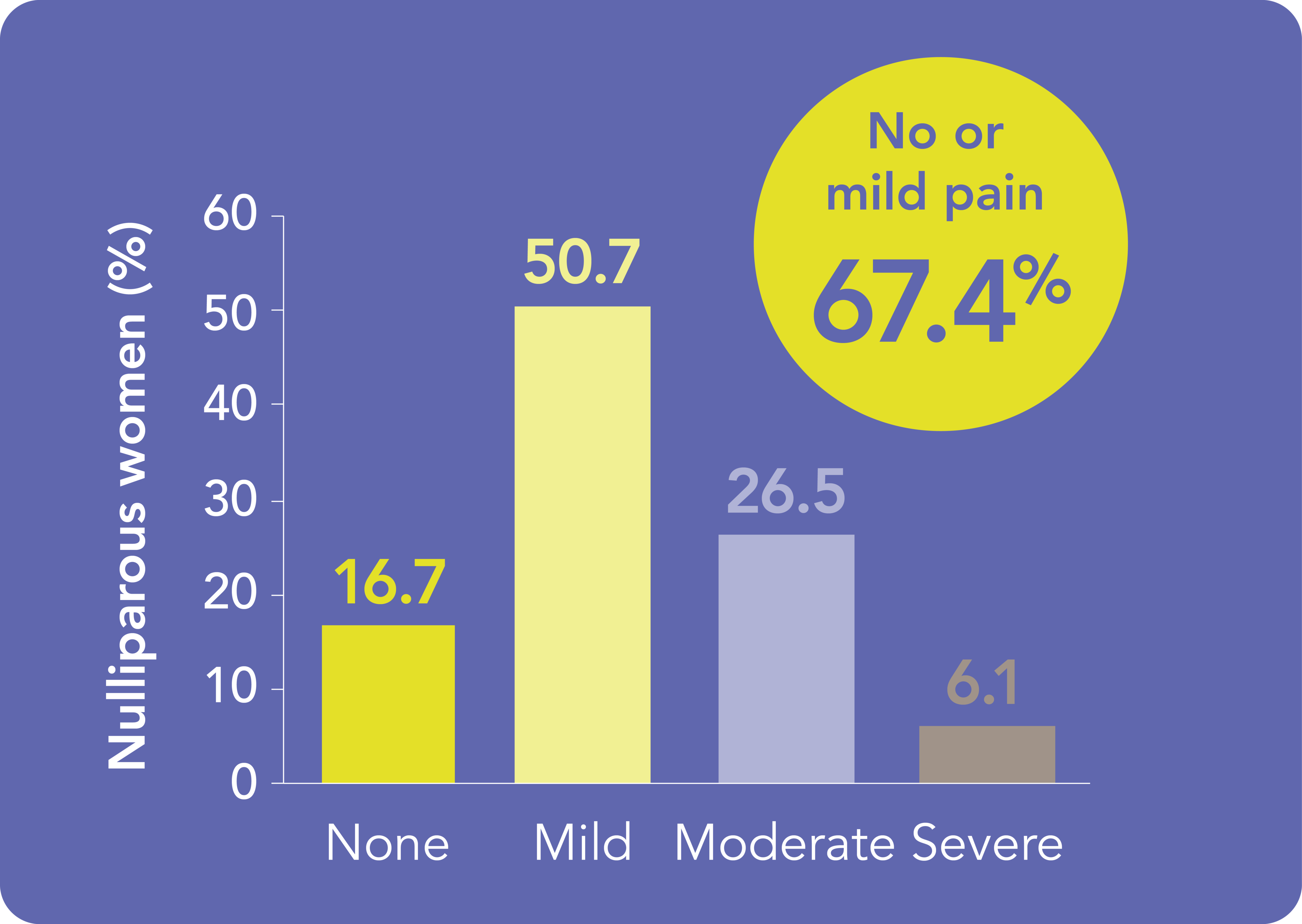
Kyleena®: a favorable placement experience for women, regardless of parity*1
Misconceptions about IUS placement may be a barrier to its use among parous and nulliparous women,2-4 but the real-world evidence from KYSS can bring confidence to your patients.1
77.7% of Kyleena® placements were associated with no or mild pain.1
Reassure both your parous and nulliparous patients that most Kyleena® placements are associated with no or mild pain1
IUS – intrauterine system; KYSS – Kyleena® Satisfaction Study
- Beckert V et al. Eur J Contracept Reprod Health Care 2020;25:182–189. Return to content
- Black K et al. Eur J Contracept Reprod Health Care 2012;17(5):340–348. Return to content
- Daniele MAS et al. Reprod Health 2017;14:119. Return to content
- Akdemir Y and Karadeniz M. Eur J Contracept Reprod Health Care 2019;24(3):240–245. Return to content
- Results from a baseline analysis of the KYSS data, conducted to evaluate demographics, ease of placement assessed by investigators, pain at placement rated by women, additional interventions for placement, and adverse events. A total of 1,110 women (574 nulliparous, 536 parous) from 7 countries were included in this analysis.1 Return to content
Title
Real-world placement experience data: now available to help you address women’s questions about Kyleena® placement
KYSS provided real-world insight into HCPs’ experience of Kyleena® placement.1
In addition, KYSS investigated patient-rated insertion pain, in both nulliparous and parous women.1
HCP – healthcare professional; KYSS – Kyleena® Satisfaction Study
- Beckert V et al. Eur J Contracept Reprod Health Care 2020;25:182–189. Return to content
Title
KYSS: study discontinuation rates1
|
SAF n, (%) |
Total |
|---|---|
|
Kyleena® still in use at planned End of Observation |
919 (81.4) |
|
Kyleena® discontinued before planned End of Observation |
210 (18.6) |
|
Discontinuation due to TEAEs |
69 (6.1) |
|
Discontinuation due to TESAEs |
4 (0.4) |
|
Primary reason for discontinuation |
|
|
Unsuccessful Kyleena® placement attempt |
3 (0.3) |
|
Lost to follow-up |
105 (9.3) |
|
Expulsion of Kyleena® |
6 (0.5) |
|
Removal of Kyleena® |
94 (8.3) |
|
– (Serious) adverse event |
62 (5.5) |
|
– Pregnancy |
3 (0.3) |
|
– Wish for pregnancy |
11 (1.0) |
|
– Switch contraceptive methods |
6 (0.5) |
|
– Dissatisfaction with Kyleena® |
9 (0.8) |
|
– Investigator decision |
1 (0.09) |
|
Not specified |
0 (0.0) |
Reassure your patients that in KYSS, Kyleena® discontinuation due to adverse events was seen in 6% of women1
KYSS – Kyleena® Satisfaction Study; SAF – safety analysis set; TEAE – treatment-emergent adverse event; TESAE – treatment-emergent serious adverse event
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content
Title
Less than 20% of women experienced adverse events with Kyleena® in KYSS1
Of 1,129 women in the SAF:1

81.5% (n=920)
reported no TEAEs
or TESAEs
16.9% (n=191)
reported a TEAE*
1.6% (n=18)
reported a TESAE
Title
Kyleena® had few discontinuations due to TEAEs and TESAEs in KYSS:
6.1 (n=69) and 0.4% (n=4) of women discontinued Kyleena® due to TEAEs and TESAEs, respectively.1
Counsel your patients on the low rates of adverse events seen with Kyleena®
- Reproductive system disorders and gastrointestinal disorders were the most common TEAEs. Return to content
KYSS – Kyleena® Satisfaction Study; SAF – safety analysis set; TEAE – treatment-emergent adverse event; TESAE – treatment-emergent serious adverse event
- Stovall DW et al. Eur J Contracept Reprod Health Care 2021 [DOI: 10.1080/13625187.2021.1975268. Online ahead of print] Return to content